DESCRIPTION
DORSAN®‘s line of polyethersulfone (PES) cartridges has been manufactured with hydrophilic and asymmetrical materials. They have been produced following all FDA food industry requirements.
The polyethersulfone (PES) features a very low level of protein retention. Thus, the DPES series is an excellent solution for beverage applications.
We produce two types of absolute PES cartridges; Single Membrane (DPES) and Double Membrane (DPESDL) to ensure perfect quality.
All cartridges are individually examined to confirm their levels of quality and integrity. The process is conducted using deionized water to reduce the level of extractables as much as possible. Our cartridges offer considerable retention capacities and flow rates.
FEATURES
10” to 40” lengths.
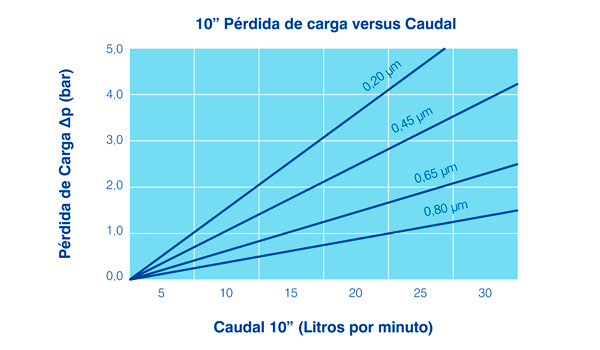
Filtration degree from 0.2 μm to 1.2 μm.
Stainless steel ring to prevent deformation.
More resistant to steam sterilization.
Available in multiple configurations.
Manufactured with no adhesives or resins.
APPLICATIONS
Wines
Mineral Waters
Beers
Fruit Juices
Pharma- Bio pharma
Api

INTEGRITY TEST*
| Pore size μm | Mín. Bubble Point 25ºC | MAx. Diffusion 25ºC |
|---|---|---|
| 0.2 | 3.4 bar | ≤16mL/min@ 2.6bar |
| 0.45 | 1.8 bar | ≤16 mL/min@ 1.4bar |
| 0.65 | 1.7 bar | ≤16 mL/min@ 1.3bar |
PLEATED POLYETHERSULFONE CARTRIDGE SPECIFICATIONS
| Absolute Rating | Porosity μm | Length” | Configurations | Joints | Inserts | |
|---|---|---|---|---|---|---|
| DPES | 0.20 | D | 10 | Blank = DOE | B = Buna-N | R = SS Insert |
| DPESDL | 0.45 | D | 20 | 1 = DOE with joints | S = Silicone | |
| DPESGF | 0.65 | D | 30 | 2 = 222 w/Flat Cap | P = Polyfoam | |
| 0.80 | D | 40 | 3 = 222 w/Fin | E = EPDM | ||
| 1.20 | 6 = 226 w/Flat | V = Viton | ||||
| 7 = 226 w/Fin | T = PTFE Encapsulated Viton | |||||
| 10 = 222 w 3 tabs/Fin |
SANITIZATION / STERILIZATION
Autokey:
124ºC, 30 min, 30 cycles
134ºC, 30 min, 20 cycles
In-Situ Steam:
124ºC, 30 min, 30 cycles, Δp <0.5 bar
134ºC, 30 min, 20 cycles, Δp <0.5 bar
MAXIMUM OPERATING CONDITIONS
Max. Operating temperature: 1.9 bar@ 82ºC
Max. Differential pressure (forward): 5.2 bar@ 25ºC
Max. Differential pressure (reverse): 2.1 bar@ 25ºC
Recommended replacement pressure: 2.4 bar
CONSTRUCTION MATERIALS
Filter Media: Asymmetric polyestersulfone (PES) membrane, Support, housing, interiors medium: Polypropylene / Fiberglass
Cartridges made with materials generally recognized as safe (GRAS) and that comply with the regulations established by the FDA (Regulation 21CFR186.1673) for use in contact with food. Health Registry # RSIPAC: 39.05248 / CAT
SECURITY
Bacterial endotoxins: <0.25 EU/mL Extractables: <40 mg/10”
DOWNLOAD PDF TECHNICAL SHEET
Note. We reserve the right to modify the information contained in this pamphlet without prior notice.
